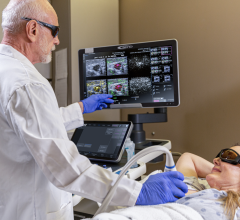
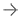August 10, 2022 — TransMed7, LLC announced that the first clinical cases of the final commercial production version of the Sparrow device (a member of the newSpeedBirdfamily of vacuum-assisted, Single Insertion / Multiple Collection (SIMC) Breast Biopsy devices) were successfully performed by Dr. Edgar Staren, a distinguished surgeon and former President of theAmerican Society of Breast Surgeons(ASBrS).
Dr. Staren commented, “Image-guided breast biopsyusing TransMed7’s SpeedBird SIMC devices involves minimal setup, is very easy to perform because of their extreme ultrasound visibility, is very well tolerated by the patients, and has taken only a few minutes to complete for each clinical case. The core specimens were simply outstanding in all the cases I performed, and provided exactly what our team needed to plan for each patient’s therapeutic management.” He added, “I can even say that these first cases spanned the spectrum of what I know to be some of the most challenging breast biopsy cases that can be encountered by physicians, and the devices performed brilliantly.”
TransMed7联合创始人兼主席James W. Vetter博士表示:“我们专门设计和开发了我们的SpeedBird和Concorde乳腺活检平台,以解决和克服与目前已有几十年历史的活检设备相关的许多问题,包括它们难以通过实时成像方式可视化针尖,尖锐且可能造成创伤的针尖,活检标本数量和质量的限制,以及它们可以收集的组织类型的限制。”
The SpeedBird and Concorde device platforms are based on TransMed7’s patented Zero5 work element composed of a fused, single element constructed from 3 hypotubes, laser cut and welded to form articulable twin cutter blades at the end of a rotating open tube. This element gently penetrates, cores, severs, and provides a pathway to transport multiple tissue samples via a closed-circuit fluid management and vacuum system into a detachable chamber. Zero5 forward coring technology enables these devices to reliably and consistently obtain full-core, uniform-diameter samples with intact architecture from all of the various soft tissues. It was developed as an entirely new mechanism intended to deliver superior results while also lowering costs, and to facilitate ease of use for rapid learning curve and adoption.
“We are delighted to announce the successful clinical utilization of our production level, minimally invasive breast biopsy devices as we formally roll out our commercial launch,” stated Dr. Vetter. “The real importance of achieving this milestone is that it demonstrates unequivocally that our Zero5 core technology has now clinically confirmed its transformational nature for the care and benefit of patients worldwide. This result proves that our plan to apply Zero5 technology to all of our next-generation biopsy device platforms, adapted for various applications all over the body, is an achievable benchmark. These include our Heron, ThunderBird, Cardinal, and Phoenix platforms among others still to come, as well as the robotic instrument applications that we derive from each of them.”
The SpeedBird platform of Breast Biopsy devices includes four models of hand-held, ultrasound guided, vacuum assisted, full fluid management, SIMC devices with simple, extremely cost-effective designs based on the common Zero5 forward coring technology with 10, 12, and 14-gauge core needle sizes. SpeedBird 38 is designed for fully automated cycling (tissue coring, part-off, and transport); SpeedBird Universal and SpeedBird Universal 99 are designed for manual coring length control and automatic tissue transport (SpeedBird Universal 99 further features a reusable handle and replaceable needles, resulting in a lower cost ideal for emerging international markets); and the new Sparrow device, which is designed for manual coring length control and automatic tissue transport and is intended to become the ultimate “Terminator” of the Single Insertion – Single Sample (SISS – limited to only one sample for each insertion) “snapgun” devices being used on a majority of patients today.
The Concorde™ platform of two device models, also based on adapted Zero5® technology, are fully automated, selectively forward coring or combined forward and shielded side coring, vacuum-assisted, full fluid management, 10-14-gauge devices. The Concorde ST device is designed for stereotactic and 2D/3D tomosynthesis guidance breast biopsy procedures with full, built-in (console-replacing) capabilities. The Concorde US device is designed for either ultrasound guidance for handheld use or with an optional, reusable, stage mount adapter for stereotactic and 2D/3D tomosynthesis use.
TransMed7联合创始人兼首席执行官Eugene H. Vetter表示:“这些设备的成功使用现在证明,我们可以适应和应用我们的专利Zero5技术,在不久的将来为活检程序和介入解决方案提供世界上最广泛的转型、下一代、微创设备平台。患者最终将能够期待下一代设备取代目前在他们身上使用的几十年前的技术。我们将在未来几周公布Concorde™系列乳腺活检设备的首个临床病例结果。”
TransMed7 anticipates participating in the next Radiological Society of North America (RSNA) meeting in Chicago, IL; the next American Society of Breast Surgeons (ASBrS) meeting in Boston, MA; and the next Society of Breast Imaging (SBI) meeting in Washington, D.C., where we will report expanded clinical results from multiple centers using our FDA-cleared SpeedBird and Concorde biopsy devices for Breast Health.
For more information:www.transmed7.com


 August 11, 2022
August 11, 2022